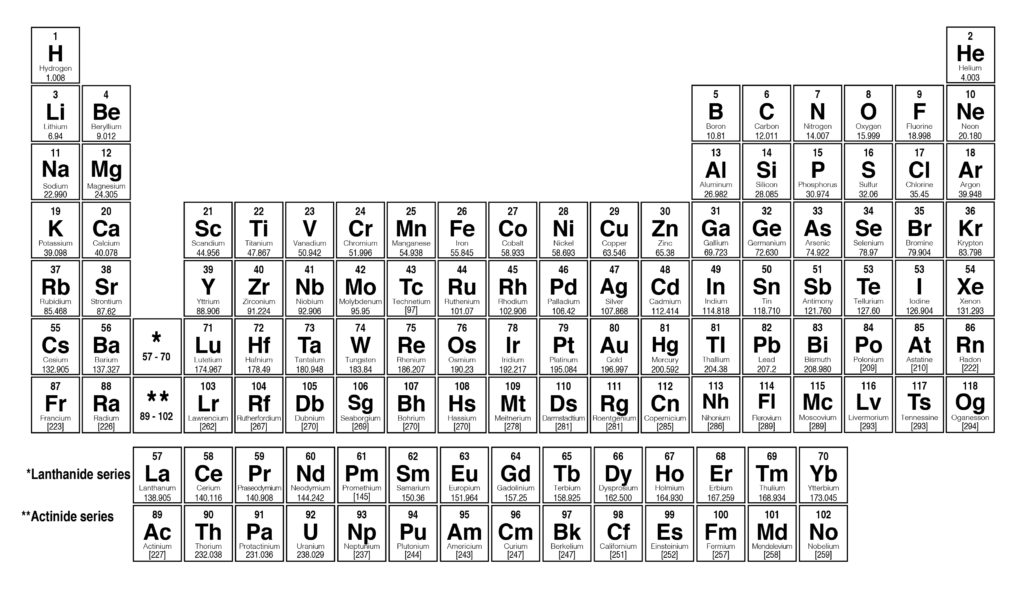
 Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole. The division sign (/) implies “per,” and “1” is implied in the denominator. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). The atomic number refers to the number of. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. Each element in the periodic table consists of atoms, and each atom has a unique atomic mass and unique atomic number. Meyer aligned the elements in his table according to periodic variations in simple atomic properties, such as atomic volume (Figure 3.4. The periodic table lists the atomic mass of carbon as 12. Visualize trends, 3D orbitals, isotopes, and mix compounds. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. 
Interactive periodic table showing names, electrons, and oxidation states.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
